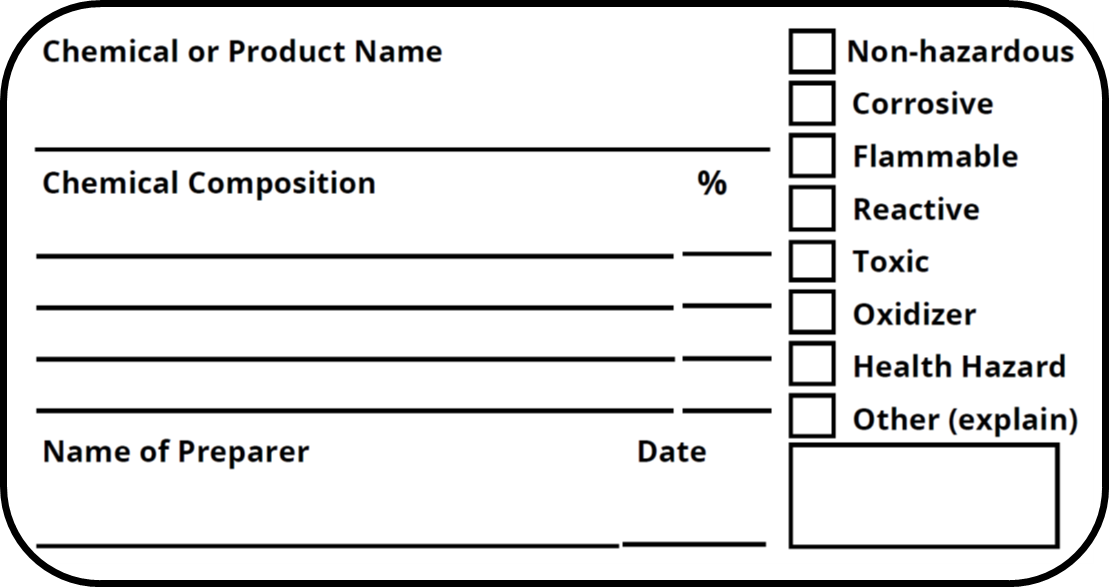
38 regulations require that product labels on containers
Shelf Life and Expiration Dating of Cosmetics | FDA There are no U.S. laws or regulations that require cosmetics to have specific shelf lives or have expiration dates on their labels. However, manufacturers are responsible for making sure their ... 1910.1030 - Bloodborne pathogens. | Occupational Safety and ... Red bags or red containers may be substituted for labels. 1910.1030(g)(1)(i)(F) Containers of blood, blood components, or blood products that are labeled as to their contents and have been released for transfusion or other clinical use are exempted from the labeling requirements of paragraph (g).
Industry Guide for the labelling of cosmetics - Canada.ca any labelling or advertising material related to a cosmetic product. The Cosmetic Regulations also prescribe the symbols and warning statements that are to be used on pressurized containers, as defined in the Consumer Chemicals and Containers Regulations, as they read on September 30, 2001. 1.2 The Consumer Packaging and Labelling Act and ...

Regulations require that product labels on containers
Canada Gazette, Part 2, Volume 156, Number 14: Regulations ... Jul 06, 2022 · Issues Labelling to improve self-selection and safe use of natural health products. In 2010-11, more than 70% of Canadians used natural health products (NHPs). footnote 3 Since then, use among Canadians has been increasing and this trend is expected to continue due to a number of factors such as Canada’s aging population, greater access to the Internet and other sources of information, and ... Changes to the Nutrition Facts Label | FDA - U.S. Food and ... Mar 07, 2022 · Manufacturers with $10 million or more in annual sales were required to update their labels by January 1, 2020; manufacturers with less than $10 million in annual food sales were required to ... Allergen labelling for food manufacturers Allergy related product withdrawals or recalls are often caused by incorrect packaging or labelling. Ensure that the correct labels are applied to products and any outer packaging. Packaging should be removed and destroyed at the end of a production run. This includes any that may be within the wrapping machine.
Regulations require that product labels on containers. Federal Register :: National Bioengineered Food Disclosure ... Dec 21, 2018 · These terms are critical in determining what foods require a BE disclosure. B. Food Subject to Disclosure. Whether a food is subject to the labeling requirements of the amended Act, depends as a preliminary matter on whether the product at issue is a food. Allergen labelling for food manufacturers Allergy related product withdrawals or recalls are often caused by incorrect packaging or labelling. Ensure that the correct labels are applied to products and any outer packaging. Packaging should be removed and destroyed at the end of a production run. This includes any that may be within the wrapping machine. Changes to the Nutrition Facts Label | FDA - U.S. Food and ... Mar 07, 2022 · Manufacturers with $10 million or more in annual sales were required to update their labels by January 1, 2020; manufacturers with less than $10 million in annual food sales were required to ... Canada Gazette, Part 2, Volume 156, Number 14: Regulations ... Jul 06, 2022 · Issues Labelling to improve self-selection and safe use of natural health products. In 2010-11, more than 70% of Canadians used natural health products (NHPs). footnote 3 Since then, use among Canadians has been increasing and this trend is expected to continue due to a number of factors such as Canada’s aging population, greater access to the Internet and other sources of information, and ...

































Post a Comment for "38 regulations require that product labels on containers"